Cathode positive or negative
The atoms join up in pairs to form Cl 2 molecules, so chlorine gas is formed at the positive electrode. Here, they lose electrons to form chlorine atoms. Negatively charged chloride ions move to the positive electrode. Here, they gain electrons to form calcium atoms, so calcium is formed at the negative electrode. Positively charged calcium ions move to the negative electrode. Predict the products of electrolysis of molten calcium chloride. Lead and bromine form during the electrolysis of molten lead bromide Worked example So lead forms at the negative electrode and bromine forms at the positive electrode. Br - ions lose electrons at the anode and become Br atoms, which pair up to form Br 2 molecules.Pb 2+ ions gain electrons at the cathode and become Pb atoms.Molten lead bromide, PbBr 2 (l), is an electrolyte. negative ions lose electrons at the positively charged anode.
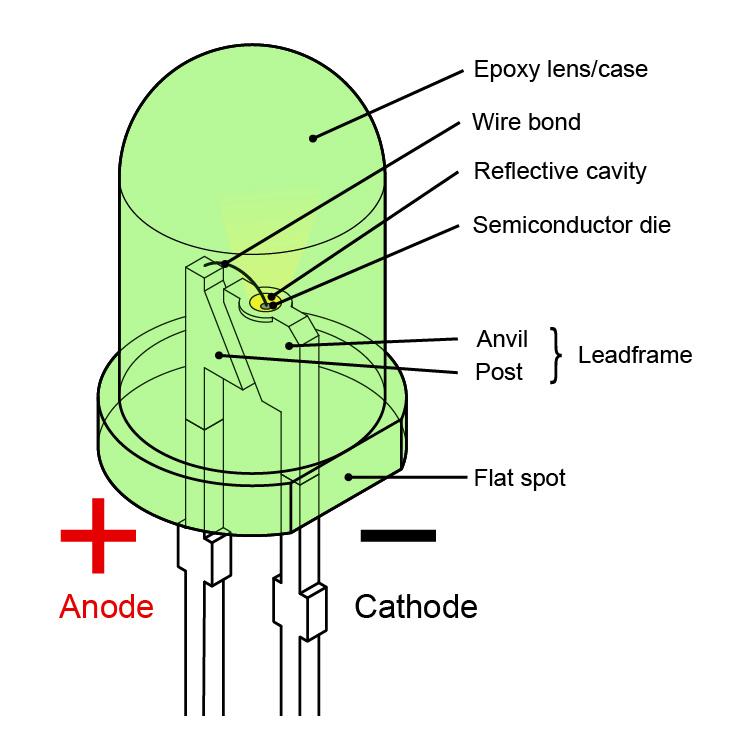
#Cathode positive or negative free#
The free moving ions in electrolytes are attracted to the oppositely charged electrodes which connect to the DC supply. Under these conditions, the ions in electrolytes are free to move within the liquid or solution.Įlectrolysis is a process in which electrical energy, from a direct current (DC) supply, breaks down electrolytes.

This will quickly wear out the battery and can also be dangerous, particularly on larger batteries. If you connect a wire between the two terminals, the electrons will flow from the negative end to the positive end as fast as they can. During charge, the positive electrode is an anode, and the negative electrode is a cathode. During discharge, the positive electrode is a cathode, and the negative electrode is an anode. The positive electrode is the electrode with a higher potential than the negative electrode. On a 9-volt or car battery, however, the terminals are situated next to each other on the top of the unit. When naming the electrodes, it is better to refer to the positive electrode and the negative electrode. In normal flashlight batteries, like AA, C or D cell, the terminals are located on the ends.

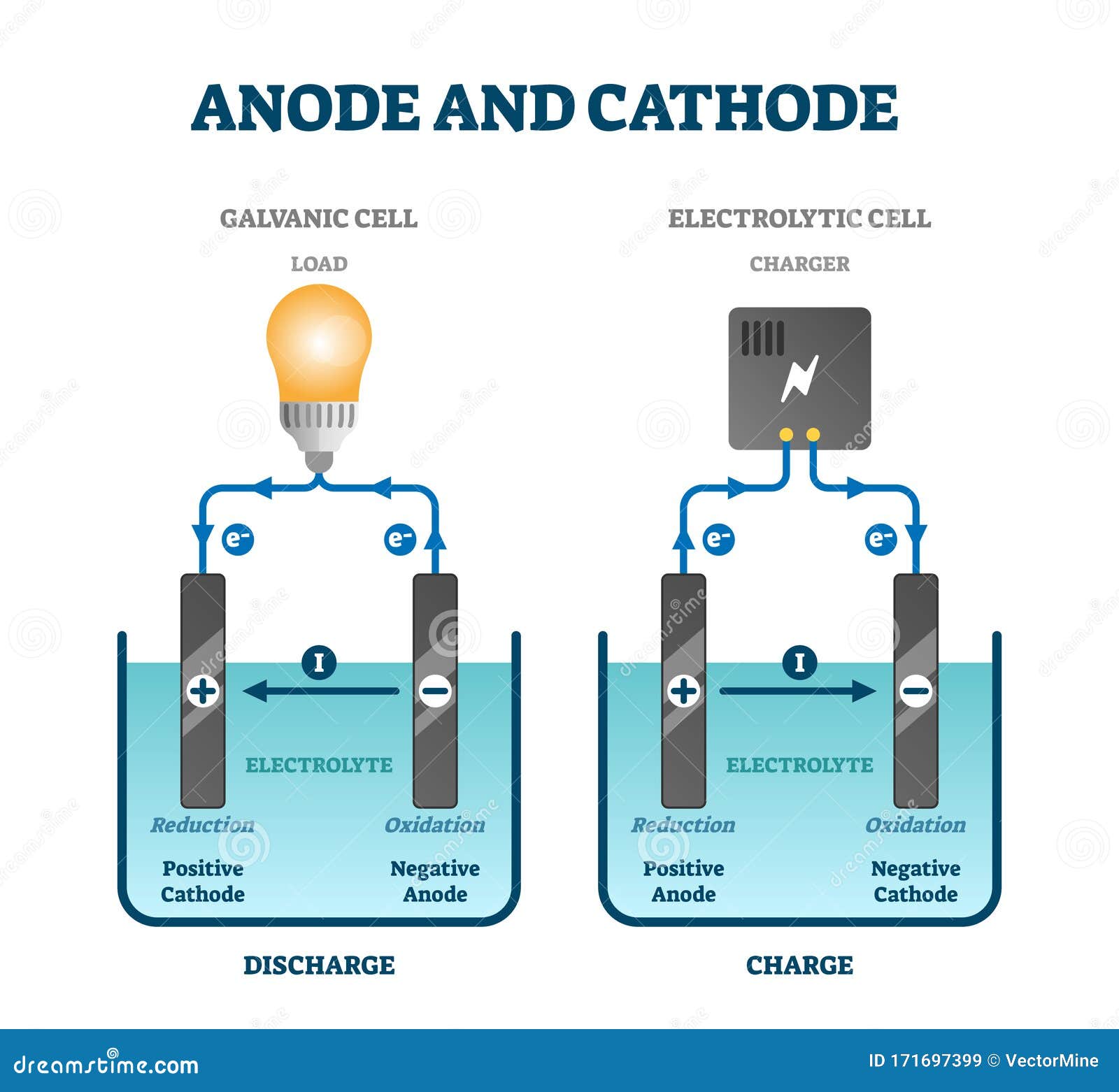
One terminal is marked (+), or positive, while the other is marked (-), or negative. Take a look at any battery, and you'll notice that it has two terminals.
